Requirements for construction of pharmaceutical cleanrooms and GMP Groups cleanroom lines for design and construction consultancy
2.1. Requirements for the construction of a pharmaceutical clean room
In the production process, the environment is the factor that most affects the quality of pharmaceutical products. Depending on the different stages of production, the level of environmental control will vary. In which all stages where the environment is exposed to the product must be strictly controlled. In order to control the environment at the production stages exposed to the product, the environment must be in a safe and quality “clean room” space.
2.2. Pharmaceutical cleanroom line designed and constructed by GMP Groups
GMP Groups advises on the design and construction of a number of pharmaceutical production lines to ensure GMP standards as follows:
– Solid drug production line: tablets, capsules, powder nudity
– Liquid drug production line: solution, mixture, emulsion.
– Semi-solid drug production line: cream, grease, putr, synamed
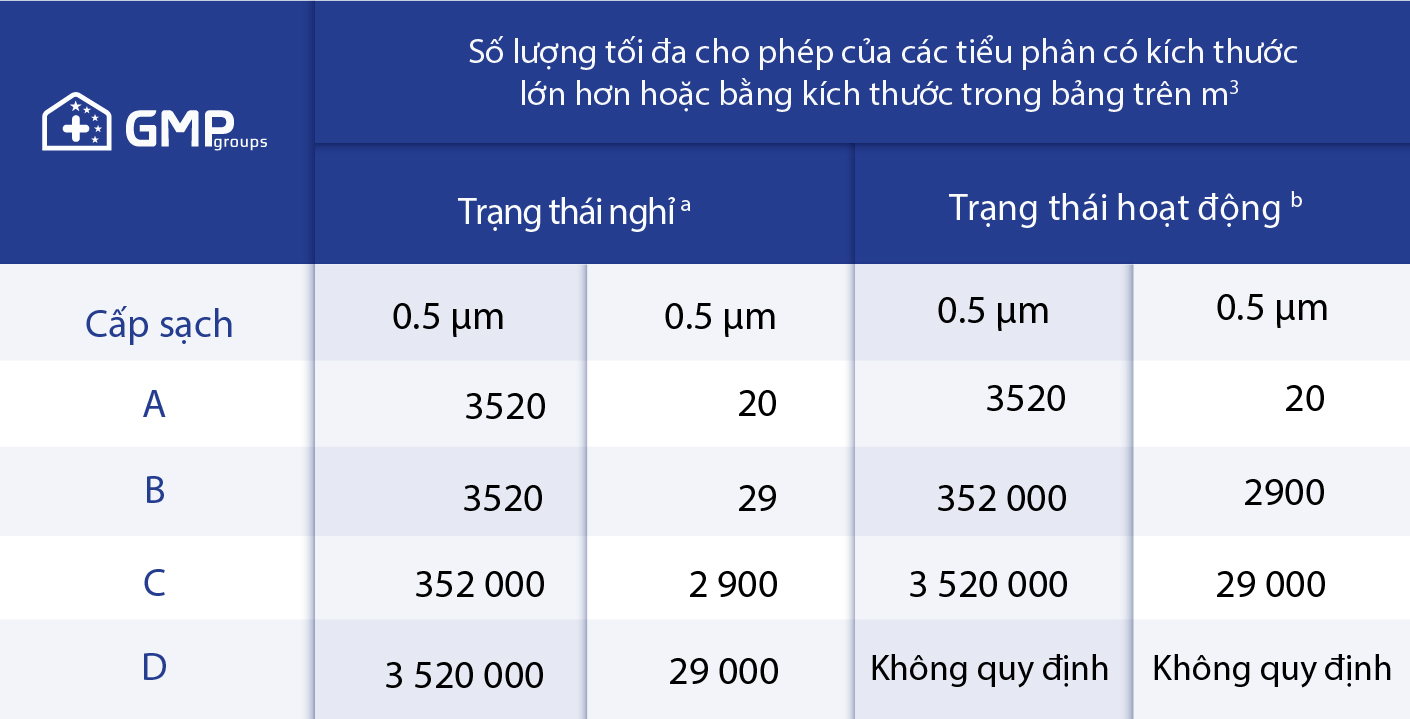
*Sources of information based on the document: WHO technical report series, No. 961, 2011 – Good practice of sterile pharmaceutical production.
Here’s an overview of GMP Groups’ pharmaceutical cleanroom service. For more information, please see more at: What is a pharmaceutical cleanroom? Standards of pharmaceutical cleanrooms
With the desire to bring a comprehensive service ecosystem related to the pharmaceutical sector, GMP Groups will be the perfect choice for any customer’s needs.
For more information on CONSULTING – DESIGN – CONSTRUCTION OF CLEANROOMS, please contact:
GMP Groups Joint Stock Company
Head office: Lot LK20.8, Ecoriver Eco-Investor, Hai Tan Ward, Hai Duong City, Hai Duong Province
Hotline: 0945255457
Website: gmpgroups.com.vn
Email: info@gmpgroups.com.vn