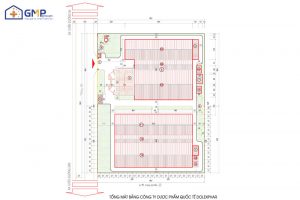
What is a veterinary cleanroom? Requirements, standards of veterinary cleanroom
Veterinary cleanrooms are a mandatory factor for veterinary pharmaceutical factories. In order for the factory to be licensed to operate production, clean rooms must meet many strict standards and requirements. So what is a veterinary clean room? What standards do veterinary medicine factories need to meet when building clean rooms? GMP Groups will answer the […]